 
Factors responsible for false reactivity in HIV immunoassays include: presence of cross-reacting antibody, inadequate centrifugation, haemolysis or lipaemia in the specimen. Use of confirmatory assays seldom resolves the problem and the HIV diagnosis remains uncertain, requiring follow up serology and molecular testing to resolve. In this setting, weakly positive HIV serology is commonly encountered. In the interest of logistic efficiency, serology is commonly performed on automated chemistry analysers in consolidated clinical pathology laboratories. They are designed to detect infection as early as possible after infection and specimens containing minute amounts of HIV antibody can test positive. Public health laboratories routinely request a second specimen in newly diagnosed patients, but frequently clinicians fail to send follow up samples.Ĥ th generation HIV immunoassays have superior sensitivity and specificity to 3 rd generation rapid tests currently in use in clinics in South Africa. Patients who are reactive in both are considered HIV-infected, in line with World Health Organisation recommendations. Initially reactive specimens are tested with a second 4 th generation immunoassay. Laboratory screening involves the use of 4 th generation antibody/p24 antigen combo immunoassays. In hospitalised patients, blood is sent to a laboratory for testing. However, ensuring a uniform quality of testing at point of care can be difficult. In the public sector, HIV testing is usually performed in clinics using rapid test devices. South Africa has the largest HIV epidemic in the world, with an estimated 5.7 million infections. Apart from this, the authors received no specific funding for this work.Ĭompeting interests: The authors have declared that no competing interests exist. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.ĭata Availability: All relevant data is included in the paper or supplementary files.įunding: The cost of reagents was covered by the Quality Assurance Division of the National Health Laboratory Service. 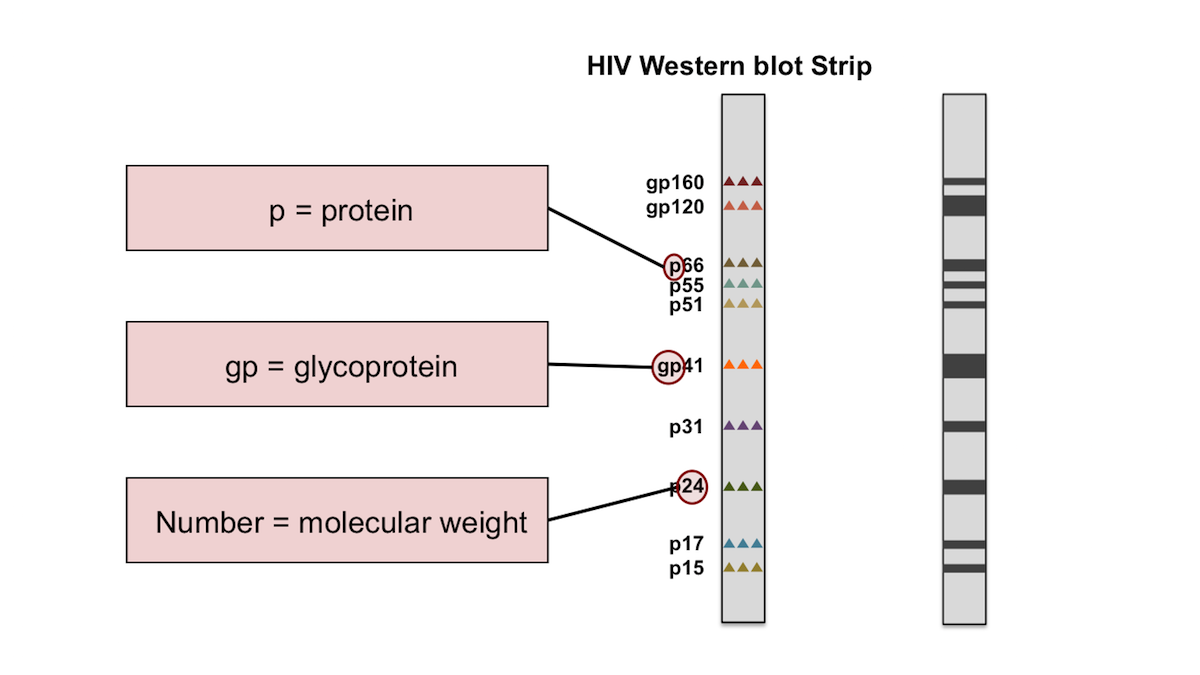
Received: DecemAccepted: JPublished: July 31, 2017Ĭopyright: © 2017 Hardie et al. Citation: Hardie DR, Korsman SN, Hsiao N-Y, Morobadi MD, Vawda S, Goedhals D (2017) Contamination with HIV antibody may be responsible for false positive results in specimens tested on automated platforms running HIV 4th generation assays in a region of high HIV prevalence. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
